- High-quality, low-cost prosthetic components
- Complete line to serve any prosthetic protocol
- Compatible with most major implant systems
- Uniform pricing regardless of system


Inclusive® Prosthetic Components
Inclusive® Prosthetic Components include a wide array of titanium abutments, along with analogs, screws, temporary and UCLA abutments, digital and conventional transfer copings, and more. Uniform pricing across systems allows for predictable restorative costs.
Increase Efficiency and Profitability of Your Implant Cases
As the world’s leading dental implant laboratory, Glidewell is proud to offer a complete line of prosthetic components manufactured by Prismatik Dentalcraft, Inc., in an ongoing effort to bring comprehensive implant treatment options to patients across the economic spectrum. Inclusive Prosthetic Components include a wide array of esthetic and multi-unit titanium abutments, along with analogs, screws, temporary and UCLA abutments, digital and conventional transfer copings, and more. Prismatik Dentalcraft has FDA 510(k) clearance for prosthetic connections that are compatible with many of the industry’s leading implant manufacturers.
Inclusive Prosthetic Components are produced in Irvine, California, in our ISO 13485-certified manufacturing facility. We ensure tight tolerances, precision machining, state-of-the-art processing and cleaning, and extensive validation testing — from fatigue strength assessment to packaging integrity analysis. Uniform pricing across systems allows for predictable restorative costs. The result is a product that offers immediate value and lasting quality.

Clinical Studies
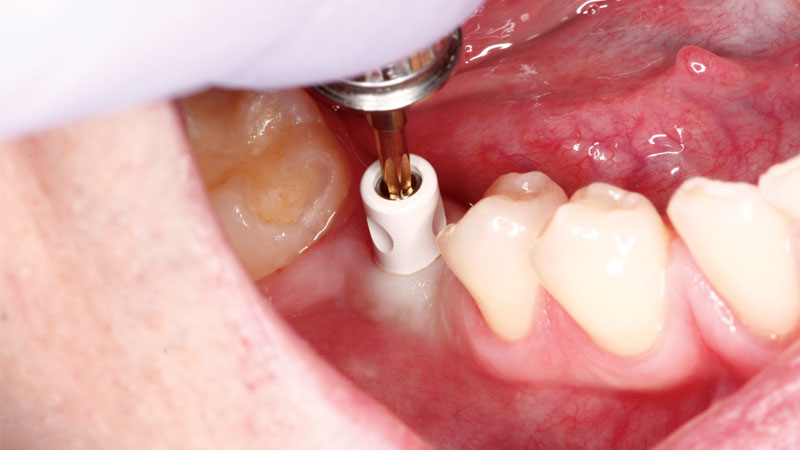
A scanning abutment was connected to the implant using a hand driver.
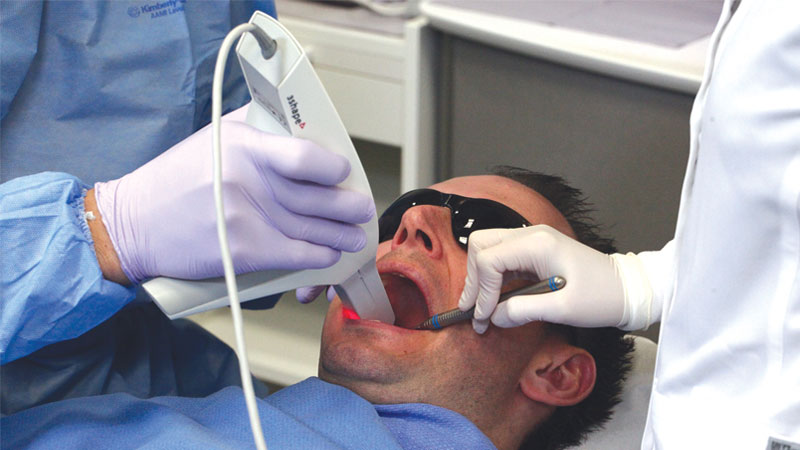
A digital final impression was taken with an intraoral scanner.
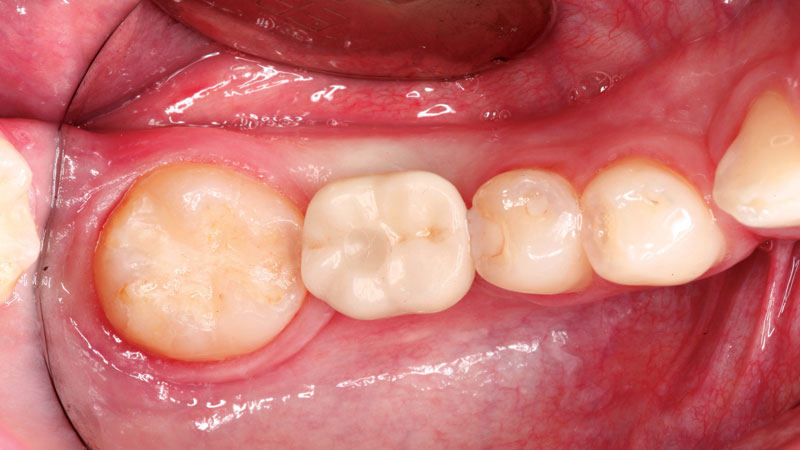
Occlusal view of the final restoration.
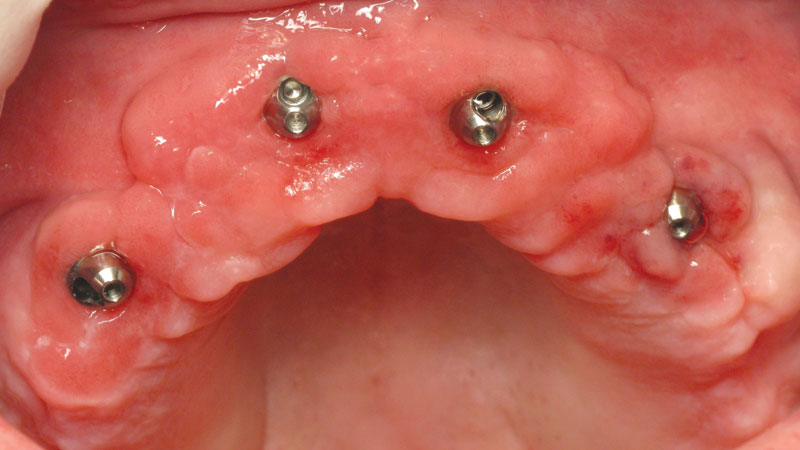
Multi-unit abutments corrected divergent implant angulation and established a uniform prosthetic platform around the arch.
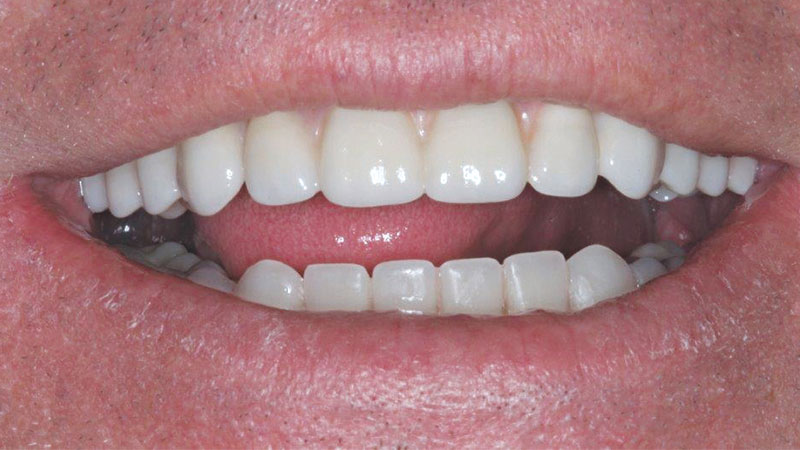
The abutments effectively addressed the unique circumstances of the case and permitted a molar-to-molar restoration.
- Slide 1
- Slide 2
Inclusive Multi-Unit Abutments were attached to the maxillary implants, correcting for the divergent angulation of the implants. This would … allow for a molar-to-molar restoration. The final BruxZir® Full-Arch Implant Prosthesis completed a dramatic oral reconstruction … restoring form, function and quality of life.
An Inclusive Scanning Abutment was threaded into the implant … The laboratory utilized the digital impression data to create ideal custom abutments. This eliminated the need to ship conventional impressions to the lab, resulting in a quicker turnaround time. The patient was pleased with the restoration, which required no adjustments.
- Slide 1
- Slide 2
Indications
Inclusive Abutments are premanufactured prosthetic components directly connected to endosseous dental implants and are intended for use as an aid in prosthetic rehabilitation.
All digitally designed abutments for use with Inclusive Abutments for CAD/CAM are intended to be manufactured at a validated milling center.
Inclusive Multi-Unit Abutments are intended to provide support and retention for multi-unit screw-retained restorations. The 30-degree multi-unit abutments must be used within 45 degrees of parallelism for a splinted restoration. The 17-degree multi-unit abutments must be used within 32 degrees of parallelism for a splinted restoration.
Material Composition
- Titanium alloy (Ti-6Al-4V ELI)
- Polyether ether ketone (PEEK)
- Polyphenylsulfone (PPSU)
- Gold alloy (60% Au, 20% Pd, 19% Pt)
- Polymers (acetal copolymer)
Implant System Compatibility
Available for BIOMET 3i™ Certain®; CAMLOG® SCREW-LINE; DENTSPLY Implants ANKYLOS® C/X, ASTRA TECH Implant System® and ASTRA TECH Implant System® EV; HIOSSEN® HG System; Inclusive® Tapered Implant System; MegaGen AnyRidge® Implant System; Nobel Biocare Brånemark System® RP, NobelActive® and NobelReplace®; Straumann® Bone Level and Tissue Level; and Zimmer Dental Screw-Vent®. Prices and availability of component types vary by system.
Inclusive is a registered trademark of Prismatik Dentalcraft, Inc. All third-party trademarks are property of their respective owners.
Pricing
|
Multi-Unit Abutment(straight or angled)
|
$116.00 |
|
Locator® Abutment
|
$133.00 |
|
Titanium Esthetic Abutment(straight or angled)
|
$95.00 |
|
Titanium Scan Body
|
$32.00 |
|
UCLA Gold Abutment
|
$116.00 |
|
UCLA Plastic Abutment
|
$39.00 |
|
Titanium Abutment(4.5 mmH, 6 mmH)
|
$37.00 |
|
Temporary Abutment
|
$32.00 |
|
Implant Analog
|
$13.00 |
|
Impression Coping
|
$32.00 |
|
Titanium Screw
|
$13.00 |
|
Titanium Screw(5-pack)
|
$53.00 |
Pricing is subject to change and does not include shipping or applicable taxes.
Policies & Warranty
Limited Warranty—Prismatik Dentalcraft, Inc. Prismatik Dentalcraft, Inc. (“Prismatik”), is the manufacturer of dental products (the “product”), including Inclusive® Dental Implants (“implants”). For a period from the original purchase date of seven (7) years for implants and six (6) months for ceramic blanks and any other product (“the warranty period”), Prismatik will at its option replace or refund the purchase price of any product, to the original purchaser (“user”), that is returned due to defects in material and manufacture.
NO GUARANTEE OR WARRANTY IS IMPLIED OTHER THAN EXPRESSLY STATED, INCLUDING ANY IMPLIED WARRANTY OF MERCHANTABILITY OR FITNESS FOR A PARTICULAR PURPOSE.
Prismatik shall not be liable for any incidental or consequential damages, whether foreseeable or not, caused by defects in the product or dental devices produced using said product. User is responsible for determining the suitability of the product for user’s application. If this product is defective within the warranty period, user’s exclusive remedy and Prismatik’s sole obligation shall be replacement or refund of the purchase price of the product. For replacement or refund under this warranty, the original purchaser shall send the product at its own expense, postage prepaid, to the seller.
Instructions for Use



